Announcement
Event Date: 2 - 4 September 2024
With the theme:
Synergy, Innovation, and Blue Economy ConservationLet's Explore Blue Economy Together
Attention to new users who are unable to log in to sabcapps.sabah.gov.my due to not receiving any verification e-mail after registration, kindly submit your access/export applications manually to sabc.sabah@gmail.com. Download the manual forms from the QR code provided.
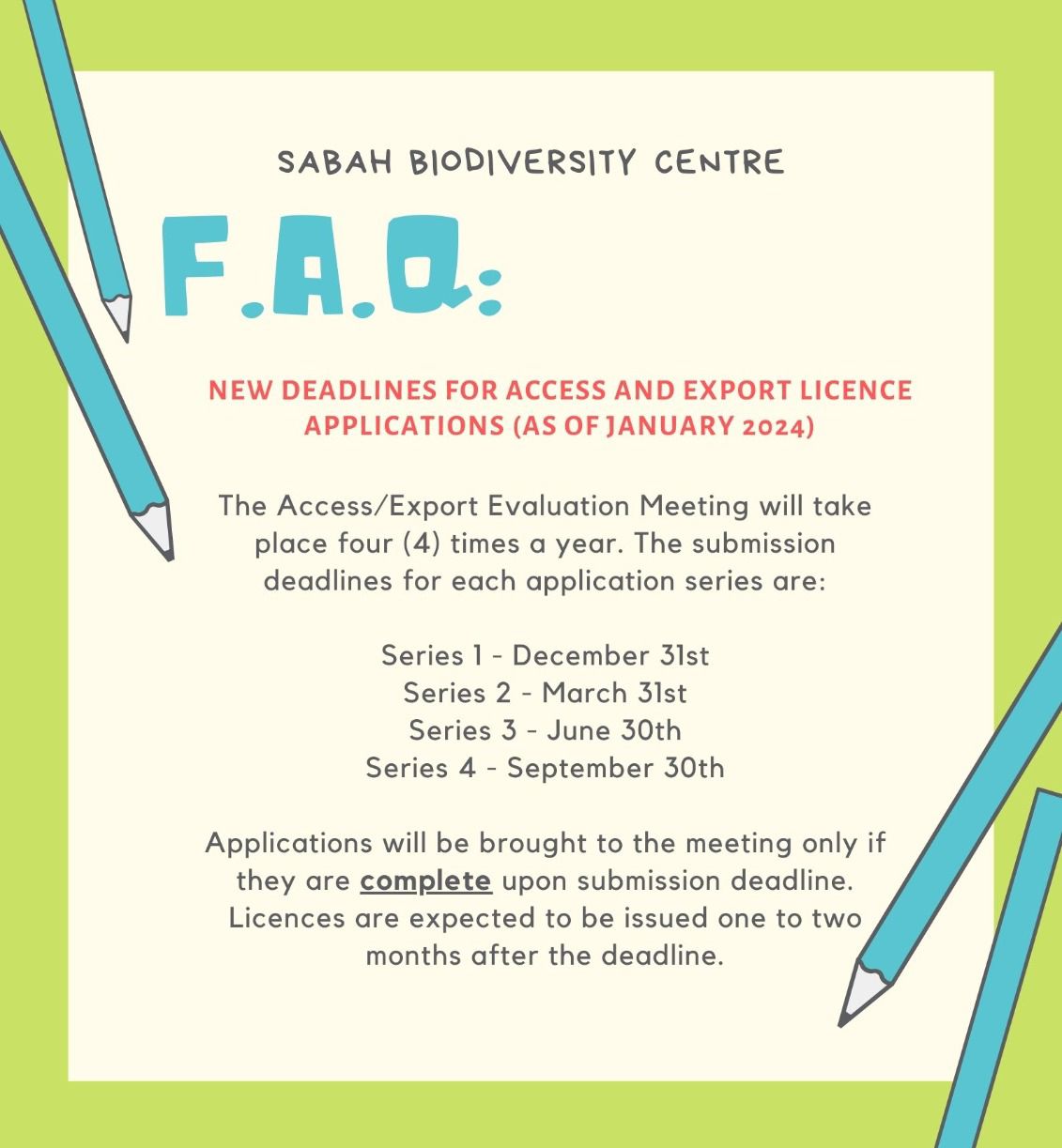
IMPORTANT ANNOUNCEMENT:
As of January 2024, the submission deadlines for the four (4) series of applications have been amended to further improve the efficiency of our service. Thank you for your cooperation.
Upcoming Events
- Konsultasi dan taklimat pengenalan kepada Projek Dokumentasi TK kepada komuniti di Kg. Sintuong-Tuong, Tambunan
- Access Export Licence Evaluation Meeting Series 2/2024
Sabah Biodiversity Enactment 2000
Know more about SBE 2000